madman
Super Moderator
Testosterone Saturation Model

 www.nature.com
www.nature.com
Abstract
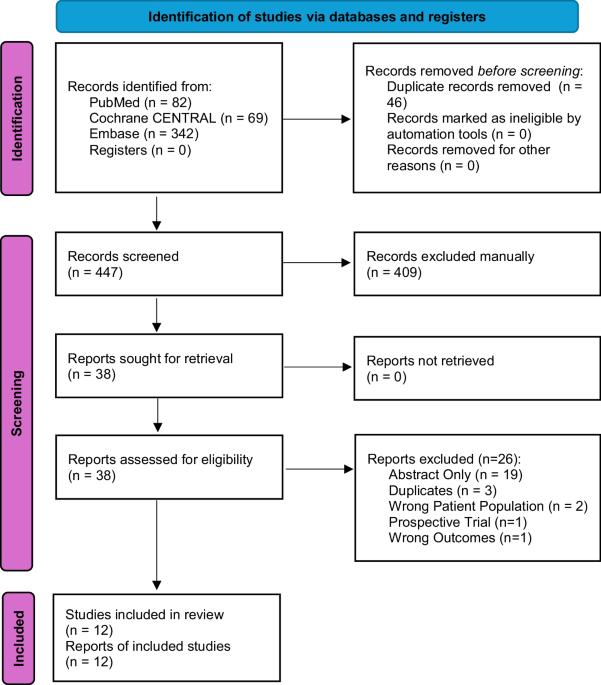
Testosterone replacement therapy (TRT) remains controversial in men with a history of prostate cancer due to historical concerns regarding oncologic safety. This scoping review aimed to systematically map existing evidence on the safety and efficacy of TRT in men following definitive treatment for prostate cancer. A systematic search of PubMed, CENTRAL, and Embase identified 447 records, from which 12 studies met inclusion criteria. Most were retrospective cohort studies, with sample sizes ranging from 10 to 152 men. TRT was not associated with an increased risk of biochemical recurrence or cancer progression in any included study. Reported PSA kinetics remained within expected post-treatment parameters, and several studies showed lower recurrence rates in TRT groups compared to controls. TRT consistently increased total and/or free testosterone and improved hypogonadal symptoms. However, the evidence base is limited by retrospective designs, small sample sizes, heterogeneous outcome reporting, and a lack of long-term data. Despite these limitations, findings suggest TRT may be cautiously considered in selected men with stable disease and confirmed hypogonadism. High-quality prospective studies are needed to clarify safety in diverse and high-risk populations and inform future clinical guidelines.
Conclusion
This scoping review systematically mapped the available evidence on TRT in men following definitive treatment for prostate cancer. Across 12 studies published between 2005 and 2025, TRT was not associated with increased risk of BCR or cancer progression. Instead, TRT demonstrated consistent efficacy in restoring serum testosterone levels and alleviating hypogonadal symptoms.
These findings challenge the historical belief that TRT poses an inherent oncologic risk to prostate cancer survivors. However, limitations in the existing evidence, such as small sample sizes, retrospective study designs, heterogeneous outcome reporting, and underrepresentation of high-risk and diverse patient groups, highlight the need for high-quality prospective research. Until such data become available, TRT may be cautiously considered in men with documented hypogonadism and stable post-treatment disease, provided that treatment decisions are individualised and closely monitored.

Testosterone replacement therapy following definitive treatment for prostate cancer: a scoping review of safety and efficacy - International Journal of Impotence Research
Testosterone replacement therapy (TRT) remains controversial in men with a history of prostate cancer due to historical concerns regarding oncologic safety. This scoping review aimed to systematically map existing evidence on the safety and efficacy of TRT in men following definitive treatment...
Abstract
Testosterone replacement therapy (TRT) remains controversial in men with a history of prostate cancer due to historical concerns regarding oncologic safety. This scoping review aimed to systematically map existing evidence on the safety and efficacy of TRT in men following definitive treatment for prostate cancer. A systematic search of PubMed, CENTRAL, and Embase identified 447 records, from which 12 studies met inclusion criteria. Most were retrospective cohort studies, with sample sizes ranging from 10 to 152 men. TRT was not associated with an increased risk of biochemical recurrence or cancer progression in any included study. Reported PSA kinetics remained within expected post-treatment parameters, and several studies showed lower recurrence rates in TRT groups compared to controls. TRT consistently increased total and/or free testosterone and improved hypogonadal symptoms. However, the evidence base is limited by retrospective designs, small sample sizes, heterogeneous outcome reporting, and a lack of long-term data. Despite these limitations, findings suggest TRT may be cautiously considered in selected men with stable disease and confirmed hypogonadism. High-quality prospective studies are needed to clarify safety in diverse and high-risk populations and inform future clinical guidelines.
Conclusion
This scoping review systematically mapped the available evidence on TRT in men following definitive treatment for prostate cancer. Across 12 studies published between 2005 and 2025, TRT was not associated with increased risk of BCR or cancer progression. Instead, TRT demonstrated consistent efficacy in restoring serum testosterone levels and alleviating hypogonadal symptoms.
These findings challenge the historical belief that TRT poses an inherent oncologic risk to prostate cancer survivors. However, limitations in the existing evidence, such as small sample sizes, retrospective study designs, heterogeneous outcome reporting, and underrepresentation of high-risk and diverse patient groups, highlight the need for high-quality prospective research. Until such data become available, TRT may be cautiously considered in men with documented hypogonadism and stable post-treatment disease, provided that treatment decisions are individualised and closely monitored.









