Nelson Vergel
Founder, ExcelMale.com
Part 1: Here
NV: Can you tell us about your experience with prescribing different options for TRT? Have you seen any difference in using gels versus injections? Is there any advantage or disadvantage to using either one?
MS: Injectable, transdermal, buccal, and oral testosterone formulations are available for clinical use. These forms of treatment differ in several key areas.
Oral preparations include methyltestosterone and fluoxymesterone, which are rarely prescribed because of their association with substantial liver toxicity. In Europe, there is an oral preparation of testosterone undecenoate, Andriol. It has a poor history of bioavailability.
The FDA approved a buccal preparation of testosterone, Striant. Striant requires administration twice per day. It is used little at this time.
Transdermal testosterone is available as a patch, Testim, and gel, Androgel. Daily application is required for each of these. They are designed to deliver 5 to 10 mg of testosterone per day. The advantages include ease of use and maintenance of relatively uniform serum testosterone levels over time. Skin irritation in the form of itching and redness is a frequent adverse effect of Testim with reports as high as 60-70%. This is uncommon with Androgel. Inadequate absorption through the skin may limit the value of transdermal preparations in some persons. A common problem is the low dose preparations provide inadequate serum testosterone levels. This is also seen with the high dose.
The topicals have become, by far, the largest used products in the last decade or so, approaching a billion dollars in sales. Androgel is the biggest product of the topicals.
If the patient is not too scared of doing injectables, oil based testosterone ester preparations are available. The most commonly used injectables are Delatestryl or testosterone enanthate and Depo-Testosterone or testosterone cypionate. In my practice, the typical dose is between 100-150 mg/week. The peak serum levels occur 2 to 5 days after injection, and a return to baseline is usually observed 10 days after injection. In this manner, adequate serum levels are maintained. Intramuscular injections of testosterone can cause local pain, soreness, bruising, redness, swelling, and possible infection.
NV: Most doctors use 1-cc of 200 milligrams every two weeks. Can you describe the problems with this schedule?
This is a problem that is seen much more often than necessary. Many doctors use a typical dose is 100 mg per week, or 200 to 300 mg every two to three weeks.
Within 7-10 days after injection, the serum testosterone levels are low to abnormally low. This is describes as a "roller coaster" effect, characterized by alternating periods of symptomatic benefit and a return to base-line symptoms, corresponding to the fluctuations in serum testosterone levels. This can be discovered by having the testosterone level checked within 24 hours prior to injection.
NV: Can you talk to us a little bit about compounding pharmacy products? I mean, particularly when using testosterone gels with concentrations higher than 1 percent for reaching total testosterone blood levels above 500 nanograms per deciliter. Have you had any experience with the compounding industry?
MS: I have had some experience with the compounding industry. The compounding pharmacies are very capable at providing higher concentrations of transdermal testosterone preparations. Because of this, they are able to supply a transdermal product in small volumes. They also tend to be less expensive than commercially available pharmaceutical testosterone replacement options.
NV: Do you think it is advisable to get your testosterone levels rechecked after a few weeks of starting any of the therapies?
MS: My protocol is that once I start a patient on testosterone, I check the testosterone level 4-6 weeks after initiating TRT. In patients using topical preparations, I recommend testing within 4-6 hours after application. Those using injectables of testosterone esters, I recommend testing within 24 hours before their next scheduled injection.
NV: Do you have any preference between the free testosterone versus total testosterone test?
MS: In the monitoring of the patient on TRT, I utilize the total testosterone. The initial evaluation of a patient might include the use of free testosterone or bioavailable testosterone. In a symptomatic individual, the total testosterone can be normal but the free or bioavailable testosterone abnormal.
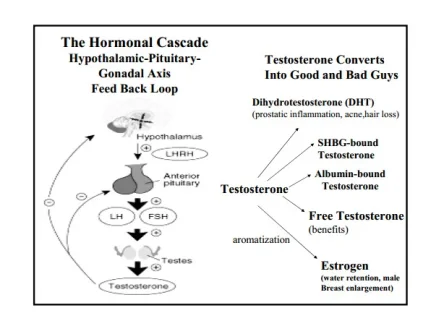
Testosterone circulates in three forms. Testosterone circulates in a free or unbound state, tightly bound to sex hormone binding globulin (SHBG), or weakly bound to the blood protein albumin. Bioavailable, non-SHBG, testosterone includes free testosterone and testosterone that is bound to albumin but does not include sex hormone binding globulin-bound testosterone.
Examined changes over time have demonstrated a decrease in total testosterone and an increase in sex hormone binding globulin (SHBG) levels. Because of this, the total testosterone might be normal whereas the free or bioavailable testosterone is abnormal. If these alternative methods are used to diagnose hypogonadism, their utility during TRT is limited.
I would caution about the assay methodology used to calculate the free or bioavailable testosterone. The methods used to conduct the measurements vary in their accuracy, standardization, the extent of validation, and the reproducibility of results.
Bioavailable testosterone is measured or calculated in several ways. SHBG bound testosterone can be precipitated with ammonium sulfate and the remaining testosterone is then taken as the bioavailable.
Measures of free testosterone (FT) are controversial. The only standardized and validated method is equilibrium dialysis or by calculating free testosterone levels based on separate measurements of testosterone and SHBG. Other measures of free testosterone are less accurate.
NV: And your goal is usually to have patients above what level?
MS: I like their total testosterone trough or lower level to be in the 500-700 range, normal being 300-1,000 ng/dL.
NV: Besides checking of the initial T level, can you elaborate on the monitoring during TRT?
I recommend periodic follow-up of patients receiving replacement testosterone therapy in intervals of 3 months during the first year of treatment. Afterwards, patents are followed-up every six months. It is important to do a review of systems to ensure the relief of the complaining symptoms as well as no worsening or new symptoms.
In addition to the serum total testosterone, I routinely monitor the basic chemistry profile, which includes liver function, kidney function, electrolytes, glucose, lipid panel, and hemoglobin or hematocrit. At three months, I will often include estradiol and dihydrotestosterone levels.
If the patient is over 50, I include the PSA. The role of digital rectal examination (DRE) and PSA in detecting early, clinically significant, prostate cancer is controversial. I discuss this with each patient and allow them to decide on their use.
NV: How about the new drugs non-steroidal androgens that are in the pipeline? Can you tell us what you've read about them?
MS: They are called SARM: selective androgen receptor modulators. They are going to become more and more popular. The closest SARM that is coming to the market, and it is years away, is called ostarine. It is being developed by GTx, Inc. Ligand Pharmaceuticals has a SARM in early phase development. They are both traded on the NASDAQ exchange.
The initial studies are being done in cancer patients. The data collected is change in muscle mass and strength. The clinical outcome being measured is the six-minute walk test.
My feeling on this is that we have a long way to go before these things come to market. If they come to market within the next 5 to 10 years, we'll be lucky. As far as I know, these are the only SARMs in human clinical trials.
NV: I've also heard that SARMs may not have any influence on sexual function, only on lean body mass and maybe some functional capacity. They are really not replacement of testosterone. Are they?
MS: From the initial studies these are meant to take the place of anabolic steroids, not testosterone. There are no indications SARMs are being developed as TRT. The data to data from both animal and human studies is that they act similarly, if not identically, to anabolic steroids. They act through the androgen receptor. They do cause HPTA suppression.
Even though they have the same effect, they will be able to be marketed without that name “anabolic steroids.” This would be an obvious advantage in their marketing. It should be noted that these drugs, SARMs, have already found their way into the nonprescription or illicit market.
NV: Can somebody on testosterone replacement become less fertile? I mean, if a man wants to impregnate his wife after, let's say, a year of testosterone replacement, is there any risk for that man to become less fertile to his wife?
MS: Testicular size and consistency often does diminish, and men should be advised that fertility would be greatly compromised during testosterone replacement therapy because of down regulation of luteinizing hormone and follicle-stimulating hormone.
The general rule is they will become less fertile. But you cannot depend on its use as a fertility drug. And that's where we come in with the contraceptive studies. We have many, many contraceptive studies that use testosterone cypionate at 200 milligrams a week and find that, yes, it decreases fertility. But there is still a subset of men that still produce sperm that are fertile.
NV: Are these men good candidates for a protocol to reset their HPGA?
MS: We had many men who came to the clinic with the actual complaint that they were using anabolic steroids, or they were using testosterone, and they now want to get their wife pregnant. Although many will return to normal after stopping TRT, this period can be lengthy.
The amazing thing to me is that the number of people that come to me, who have seen the doctor, who are either non-prescription or prescription anabolic steroid users, testosterone with or without combination of anabolic steroids, who have the problem of infertility; and their doctors have no idea what to do, except to do nothing. But on top of this are all the psychological problems and effects that come along with doing nothing as a consequence of anabolic steroid induced hypogonadism. The HPTA protocol has restored fertility as well as decreased the time substantially.
NV: Can you tell us briefly about the concept of resetting the HPGA axis?

MS: There are clear conditions under which testosterone requires administration for life-long treatment. However, there are increasing numbers of individuals being treated with TRT who do not fall under these disorders. TRT is more and more being prescribed for late onset hypogonadism. This is called many other names, including andropause, androgen deficiency of the aging male, and others.
There are no consequences of the decline in serum testosterone with age that are known with certainty. Several parallels exist between the effects of aging and those of hypogonadism, which suggest that the decline in serum testosterone might be a cause of several effects of aging. Since the long-term effects of androgen treatment for late onset hypogonadism or andropause are not well-known, I discontinue therapy on an approximate annual basis to ensure HPTA normalization—functionality. This allows the patient the autonomy to stop therapy should the need arise.
What is clear is that upon stopping testosterone or anabolic steroids, a period of anabolic steroid induced hypogonadism ensues. This occurs in one hundred percent of individuals stopping testosterone. The only variables are the duration and severity. The duration of the hypogonadism, or the severity of the hypogonadism, is typically related to the anabolic steroid drug, dose, and duration.
In other words, one person that is on testosterone for an entire year; they may come back to normal within one or two months. Another person may take 12, 18 months, or even three years to come back to normal. The best studies we have on this are the contraceptive studies with testosterone for over a year. And what we find in those studies is that it may take up to three years for a person to return to normal.
If they have been taking those anabolic steroids to improve their body composition, increase the lean body mass and decrease the body fat, that all goes back to normal after stopping the anabolic steroids. But you are also going to be exposed to the other adverse effects of hypogonadism, which include adverse psychological and cardiovascular effects. Some of the adverse psychological effects are depression, decreased cognitive abilities, insomnia, decreased libido, and erection dysfunction. More significantly, after cessation adds the comorbid condition of hypogonadism to their already existing chronic illness.
NV: There is some data on hypogonadism and increased risks of cardiovascular events. Is that what you mean? That since people actually become severely hypogonadal, they actually may be risking higher incidence of heart attacks and strokes?
MS: That is correct. There are numerous studies demonstrating the relationship between low testosterone levels and adverse cardiovascular events, as well as stroke. Also, there are case study reports of people who stop anabolic steroids, and then suffer a heart attack.
In the book that I wrote, one of the studies in the published literature, looking at the effects of anabolic steroids in certain populations, for 12 weeks, did not look at the patients after they stopped the drug. If you want to look at the effects of these drugs, you need to see what happens when you stop them. I filed a Freedom of Information Act to obtain the patient records. One of the patients actually suffered a heart attack within four weeks of stopping the anabolic steroid. The details, including the original patient records, of this case are reported in my book.
NV: Can you expand about resetting the HPGA?
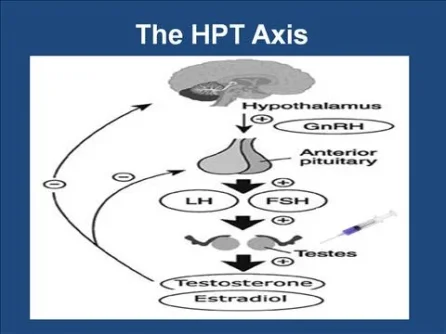
The word resetting is a misnomer, although recent studies published in the New England Journal of Medicine do indicate this possible. In 2007, the NEJM reports on the resetting of the HPTA after TRT for adult onset idiopathic hypogonadism. This is the first report demonstrating HPTA plasticity in adulthood. The term I prefer is HPTA functionality and restoration.
AAS, including testosterone, licit and illicit, administration induce a state of hypogonadism that continues after their cessation. All compounds classified as androgens or anabolic steroids cause a negative feedback inhibition of the hypothalamic pituitary testicular axis, suppress endogenous gonadotropin secretion, and as a consequence serum testosterone.
The symptoms of AIH are identical to classical hypogonadism. This problem prevents many of discontinuing testosterone or anabolic steroids. As we have said, there are many reasons for stopping testosterone, including polycythemia, gynecomastia, and other issue as compliance, affordability, and changing life style.
The accepted standard of care within the medical community for anabolic steroid induced hypogonadism is to do nothing with the expectation the individual will return to normal unassisted. But the literature shows this not to be the case.
AIH is critical towards any future planned use of AAS or similar compound to effect positive changes in muscle mass and muscle strength as well as an understanding for what has been termed anabolic steroid dependency. The further understanding and treatments that mitigate or prevent AIH could contribute to androgen therapies for wasting associated diseases and stopping nonprescription AAS use.
NV: What is used for this resetting of the hormonal axis?
MS: A combination of three drugs. The individual use of hCG, clomiphene citrate, and tamoxifen are well-known, well-accepted, and well-tested standards of care treatments in peer-reviewed medical literature for the diagnostic testing for underlying pathology of hypogonadism. The HPTA protocol uses the medications human chorionic gonadotropin -hCG, clomiphene citrate, and tamoxifen.
The first phase of the HPTA protocol examines the functionality of the testicles by the direct action of hCG. hCG raises sex hormone levels directly through the stimulation of testis and secondarily decreases the production and level of the gonadotropin LH. The increase in serum testosterone with the hCG stimulation is useful in determining whether any primary testicular dysfunction is present.
This initial value is a measure of the ability of the testicles to respond to stimulation from the hCG. Demonstration of HPTA functionality is by an adequate response of the testicles to raise the serum level of T well into the normal range. If this is observed the hCG is discontinued. The failure of the testes to respond to an hCG challenge is indicative of primary testicular failure. In the simplest terms, the first half of the protocol is determine testicular production and reserve by direct stimulation with hCG. If one is unable to obtain adequate (normal) levels successfully to the first half there is little cause or reason to proceed to the second half.
The second phase of the HPTA protocol, clomiphene and tamoxifen, examines the ability of the hypothalamo-pituitary to respond to stimulation by producing LH levels within the normal reference range. The clomiphene citrate challenge differentiates secondary hypogonadism. Clomiphene is an antiestrogen, which decreases the estrogen effect in the body. It has a dual effect by stimulating the hypothalamic pituitary area and it has an antiestrogenic effect, so that it decreases the effect of estrogen in the body. Tamoxifen is more of a strict antiestrogen, it decreases the effect of estrogen in the body, and potentiates the action of clomiphene. Tamoxifen and clomiphene citrate compete with estrogen for estrogen receptor binding sites, thus eliminating excess estrogen circulation at the level of the hypothalamus and pituitary, allowing gonadotropin production to resume.
Administration produces an elevation of LH and secondarily gonadal sex hormones. The administration of clomiphene leads to an appropriate rise in the levels of LH, suggesting that the negative feedback control on the hypothalamus is intact and that the storage and release of gonadotropins by the pituitary is normal. If there was a successful stimulation of testicular T levels by hCG, but an inadequate or no response in LH production, then the patient has hypogonadotropic, secondary, hypogonadism.
In the simplest terms, the second half of the protocol is to determine hypothalamo-pituitary production and reserve with clomiphene and tamoxifen. The physiological type of hypogonadism—hypogonadotropic or secondary—is characterized by abnormal low or low normal gonadotropin (LH) production in response to clomiphene citrate and tamoxifen. In the functional type of hypogonadism, the ability to stimulate the HPTA to produce LH and T levels within the normal reference range occurs.
There is a dearth of good studies in anabolic steroids, both while you're taking them and after you stop them, I think this is going to be something that we're going to need to look at in the future. In fact, we are going to plan on looking at it in our proposed clinical studies that we have with our company for the prevention of anabolic steroid-induced hypogonadism.
Subscribe to Dr Scally's blog: http://michaelscally.blogspot.com/
More on HPTA reset
NV: Can you tell us about your experience with prescribing different options for TRT? Have you seen any difference in using gels versus injections? Is there any advantage or disadvantage to using either one?
MS: Injectable, transdermal, buccal, and oral testosterone formulations are available for clinical use. These forms of treatment differ in several key areas.
Oral preparations include methyltestosterone and fluoxymesterone, which are rarely prescribed because of their association with substantial liver toxicity. In Europe, there is an oral preparation of testosterone undecenoate, Andriol. It has a poor history of bioavailability.
The FDA approved a buccal preparation of testosterone, Striant. Striant requires administration twice per day. It is used little at this time.
Transdermal testosterone is available as a patch, Testim, and gel, Androgel. Daily application is required for each of these. They are designed to deliver 5 to 10 mg of testosterone per day. The advantages include ease of use and maintenance of relatively uniform serum testosterone levels over time. Skin irritation in the form of itching and redness is a frequent adverse effect of Testim with reports as high as 60-70%. This is uncommon with Androgel. Inadequate absorption through the skin may limit the value of transdermal preparations in some persons. A common problem is the low dose preparations provide inadequate serum testosterone levels. This is also seen with the high dose.
The topicals have become, by far, the largest used products in the last decade or so, approaching a billion dollars in sales. Androgel is the biggest product of the topicals.
If the patient is not too scared of doing injectables, oil based testosterone ester preparations are available. The most commonly used injectables are Delatestryl or testosterone enanthate and Depo-Testosterone or testosterone cypionate. In my practice, the typical dose is between 100-150 mg/week. The peak serum levels occur 2 to 5 days after injection, and a return to baseline is usually observed 10 days after injection. In this manner, adequate serum levels are maintained. Intramuscular injections of testosterone can cause local pain, soreness, bruising, redness, swelling, and possible infection.
NV: Most doctors use 1-cc of 200 milligrams every two weeks. Can you describe the problems with this schedule?
This is a problem that is seen much more often than necessary. Many doctors use a typical dose is 100 mg per week, or 200 to 300 mg every two to three weeks.
Within 7-10 days after injection, the serum testosterone levels are low to abnormally low. This is describes as a "roller coaster" effect, characterized by alternating periods of symptomatic benefit and a return to base-line symptoms, corresponding to the fluctuations in serum testosterone levels. This can be discovered by having the testosterone level checked within 24 hours prior to injection.
NV: Can you talk to us a little bit about compounding pharmacy products? I mean, particularly when using testosterone gels with concentrations higher than 1 percent for reaching total testosterone blood levels above 500 nanograms per deciliter. Have you had any experience with the compounding industry?
MS: I have had some experience with the compounding industry. The compounding pharmacies are very capable at providing higher concentrations of transdermal testosterone preparations. Because of this, they are able to supply a transdermal product in small volumes. They also tend to be less expensive than commercially available pharmaceutical testosterone replacement options.
NV: Do you think it is advisable to get your testosterone levels rechecked after a few weeks of starting any of the therapies?
MS: My protocol is that once I start a patient on testosterone, I check the testosterone level 4-6 weeks after initiating TRT. In patients using topical preparations, I recommend testing within 4-6 hours after application. Those using injectables of testosterone esters, I recommend testing within 24 hours before their next scheduled injection.
NV: Do you have any preference between the free testosterone versus total testosterone test?
MS: In the monitoring of the patient on TRT, I utilize the total testosterone. The initial evaluation of a patient might include the use of free testosterone or bioavailable testosterone. In a symptomatic individual, the total testosterone can be normal but the free or bioavailable testosterone abnormal.
Testosterone circulates in three forms. Testosterone circulates in a free or unbound state, tightly bound to sex hormone binding globulin (SHBG), or weakly bound to the blood protein albumin. Bioavailable, non-SHBG, testosterone includes free testosterone and testosterone that is bound to albumin but does not include sex hormone binding globulin-bound testosterone.
Examined changes over time have demonstrated a decrease in total testosterone and an increase in sex hormone binding globulin (SHBG) levels. Because of this, the total testosterone might be normal whereas the free or bioavailable testosterone is abnormal. If these alternative methods are used to diagnose hypogonadism, their utility during TRT is limited.
I would caution about the assay methodology used to calculate the free or bioavailable testosterone. The methods used to conduct the measurements vary in their accuracy, standardization, the extent of validation, and the reproducibility of results.
Bioavailable testosterone is measured or calculated in several ways. SHBG bound testosterone can be precipitated with ammonium sulfate and the remaining testosterone is then taken as the bioavailable.
Measures of free testosterone (FT) are controversial. The only standardized and validated method is equilibrium dialysis or by calculating free testosterone levels based on separate measurements of testosterone and SHBG. Other measures of free testosterone are less accurate.
NV: And your goal is usually to have patients above what level?
MS: I like their total testosterone trough or lower level to be in the 500-700 range, normal being 300-1,000 ng/dL.
NV: Besides checking of the initial T level, can you elaborate on the monitoring during TRT?
I recommend periodic follow-up of patients receiving replacement testosterone therapy in intervals of 3 months during the first year of treatment. Afterwards, patents are followed-up every six months. It is important to do a review of systems to ensure the relief of the complaining symptoms as well as no worsening or new symptoms.
In addition to the serum total testosterone, I routinely monitor the basic chemistry profile, which includes liver function, kidney function, electrolytes, glucose, lipid panel, and hemoglobin or hematocrit. At three months, I will often include estradiol and dihydrotestosterone levels.
If the patient is over 50, I include the PSA. The role of digital rectal examination (DRE) and PSA in detecting early, clinically significant, prostate cancer is controversial. I discuss this with each patient and allow them to decide on their use.
NV: How about the new drugs non-steroidal androgens that are in the pipeline? Can you tell us what you've read about them?
MS: They are called SARM: selective androgen receptor modulators. They are going to become more and more popular. The closest SARM that is coming to the market, and it is years away, is called ostarine. It is being developed by GTx, Inc. Ligand Pharmaceuticals has a SARM in early phase development. They are both traded on the NASDAQ exchange.
The initial studies are being done in cancer patients. The data collected is change in muscle mass and strength. The clinical outcome being measured is the six-minute walk test.
My feeling on this is that we have a long way to go before these things come to market. If they come to market within the next 5 to 10 years, we'll be lucky. As far as I know, these are the only SARMs in human clinical trials.
NV: I've also heard that SARMs may not have any influence on sexual function, only on lean body mass and maybe some functional capacity. They are really not replacement of testosterone. Are they?
MS: From the initial studies these are meant to take the place of anabolic steroids, not testosterone. There are no indications SARMs are being developed as TRT. The data to data from both animal and human studies is that they act similarly, if not identically, to anabolic steroids. They act through the androgen receptor. They do cause HPTA suppression.
Even though they have the same effect, they will be able to be marketed without that name “anabolic steroids.” This would be an obvious advantage in their marketing. It should be noted that these drugs, SARMs, have already found their way into the nonprescription or illicit market.
NV: Can somebody on testosterone replacement become less fertile? I mean, if a man wants to impregnate his wife after, let's say, a year of testosterone replacement, is there any risk for that man to become less fertile to his wife?
MS: Testicular size and consistency often does diminish, and men should be advised that fertility would be greatly compromised during testosterone replacement therapy because of down regulation of luteinizing hormone and follicle-stimulating hormone.
The general rule is they will become less fertile. But you cannot depend on its use as a fertility drug. And that's where we come in with the contraceptive studies. We have many, many contraceptive studies that use testosterone cypionate at 200 milligrams a week and find that, yes, it decreases fertility. But there is still a subset of men that still produce sperm that are fertile.
NV: Are these men good candidates for a protocol to reset their HPGA?
MS: We had many men who came to the clinic with the actual complaint that they were using anabolic steroids, or they were using testosterone, and they now want to get their wife pregnant. Although many will return to normal after stopping TRT, this period can be lengthy.
The amazing thing to me is that the number of people that come to me, who have seen the doctor, who are either non-prescription or prescription anabolic steroid users, testosterone with or without combination of anabolic steroids, who have the problem of infertility; and their doctors have no idea what to do, except to do nothing. But on top of this are all the psychological problems and effects that come along with doing nothing as a consequence of anabolic steroid induced hypogonadism. The HPTA protocol has restored fertility as well as decreased the time substantially.
NV: Can you tell us briefly about the concept of resetting the HPGA axis?
MS: There are clear conditions under which testosterone requires administration for life-long treatment. However, there are increasing numbers of individuals being treated with TRT who do not fall under these disorders. TRT is more and more being prescribed for late onset hypogonadism. This is called many other names, including andropause, androgen deficiency of the aging male, and others.
There are no consequences of the decline in serum testosterone with age that are known with certainty. Several parallels exist between the effects of aging and those of hypogonadism, which suggest that the decline in serum testosterone might be a cause of several effects of aging. Since the long-term effects of androgen treatment for late onset hypogonadism or andropause are not well-known, I discontinue therapy on an approximate annual basis to ensure HPTA normalization—functionality. This allows the patient the autonomy to stop therapy should the need arise.
What is clear is that upon stopping testosterone or anabolic steroids, a period of anabolic steroid induced hypogonadism ensues. This occurs in one hundred percent of individuals stopping testosterone. The only variables are the duration and severity. The duration of the hypogonadism, or the severity of the hypogonadism, is typically related to the anabolic steroid drug, dose, and duration.
In other words, one person that is on testosterone for an entire year; they may come back to normal within one or two months. Another person may take 12, 18 months, or even three years to come back to normal. The best studies we have on this are the contraceptive studies with testosterone for over a year. And what we find in those studies is that it may take up to three years for a person to return to normal.
If they have been taking those anabolic steroids to improve their body composition, increase the lean body mass and decrease the body fat, that all goes back to normal after stopping the anabolic steroids. But you are also going to be exposed to the other adverse effects of hypogonadism, which include adverse psychological and cardiovascular effects. Some of the adverse psychological effects are depression, decreased cognitive abilities, insomnia, decreased libido, and erection dysfunction. More significantly, after cessation adds the comorbid condition of hypogonadism to their already existing chronic illness.
NV: There is some data on hypogonadism and increased risks of cardiovascular events. Is that what you mean? That since people actually become severely hypogonadal, they actually may be risking higher incidence of heart attacks and strokes?
MS: That is correct. There are numerous studies demonstrating the relationship between low testosterone levels and adverse cardiovascular events, as well as stroke. Also, there are case study reports of people who stop anabolic steroids, and then suffer a heart attack.
In the book that I wrote, one of the studies in the published literature, looking at the effects of anabolic steroids in certain populations, for 12 weeks, did not look at the patients after they stopped the drug. If you want to look at the effects of these drugs, you need to see what happens when you stop them. I filed a Freedom of Information Act to obtain the patient records. One of the patients actually suffered a heart attack within four weeks of stopping the anabolic steroid. The details, including the original patient records, of this case are reported in my book.
NV: Can you expand about resetting the HPGA?
The word resetting is a misnomer, although recent studies published in the New England Journal of Medicine do indicate this possible. In 2007, the NEJM reports on the resetting of the HPTA after TRT for adult onset idiopathic hypogonadism. This is the first report demonstrating HPTA plasticity in adulthood. The term I prefer is HPTA functionality and restoration.
AAS, including testosterone, licit and illicit, administration induce a state of hypogonadism that continues after their cessation. All compounds classified as androgens or anabolic steroids cause a negative feedback inhibition of the hypothalamic pituitary testicular axis, suppress endogenous gonadotropin secretion, and as a consequence serum testosterone.
The symptoms of AIH are identical to classical hypogonadism. This problem prevents many of discontinuing testosterone or anabolic steroids. As we have said, there are many reasons for stopping testosterone, including polycythemia, gynecomastia, and other issue as compliance, affordability, and changing life style.
The accepted standard of care within the medical community for anabolic steroid induced hypogonadism is to do nothing with the expectation the individual will return to normal unassisted. But the literature shows this not to be the case.
AIH is critical towards any future planned use of AAS or similar compound to effect positive changes in muscle mass and muscle strength as well as an understanding for what has been termed anabolic steroid dependency. The further understanding and treatments that mitigate or prevent AIH could contribute to androgen therapies for wasting associated diseases and stopping nonprescription AAS use.
NV: What is used for this resetting of the hormonal axis?
MS: A combination of three drugs. The individual use of hCG, clomiphene citrate, and tamoxifen are well-known, well-accepted, and well-tested standards of care treatments in peer-reviewed medical literature for the diagnostic testing for underlying pathology of hypogonadism. The HPTA protocol uses the medications human chorionic gonadotropin -hCG, clomiphene citrate, and tamoxifen.
The first phase of the HPTA protocol examines the functionality of the testicles by the direct action of hCG. hCG raises sex hormone levels directly through the stimulation of testis and secondarily decreases the production and level of the gonadotropin LH. The increase in serum testosterone with the hCG stimulation is useful in determining whether any primary testicular dysfunction is present.
This initial value is a measure of the ability of the testicles to respond to stimulation from the hCG. Demonstration of HPTA functionality is by an adequate response of the testicles to raise the serum level of T well into the normal range. If this is observed the hCG is discontinued. The failure of the testes to respond to an hCG challenge is indicative of primary testicular failure. In the simplest terms, the first half of the protocol is determine testicular production and reserve by direct stimulation with hCG. If one is unable to obtain adequate (normal) levels successfully to the first half there is little cause or reason to proceed to the second half.
The second phase of the HPTA protocol, clomiphene and tamoxifen, examines the ability of the hypothalamo-pituitary to respond to stimulation by producing LH levels within the normal reference range. The clomiphene citrate challenge differentiates secondary hypogonadism. Clomiphene is an antiestrogen, which decreases the estrogen effect in the body. It has a dual effect by stimulating the hypothalamic pituitary area and it has an antiestrogenic effect, so that it decreases the effect of estrogen in the body. Tamoxifen is more of a strict antiestrogen, it decreases the effect of estrogen in the body, and potentiates the action of clomiphene. Tamoxifen and clomiphene citrate compete with estrogen for estrogen receptor binding sites, thus eliminating excess estrogen circulation at the level of the hypothalamus and pituitary, allowing gonadotropin production to resume.
Administration produces an elevation of LH and secondarily gonadal sex hormones. The administration of clomiphene leads to an appropriate rise in the levels of LH, suggesting that the negative feedback control on the hypothalamus is intact and that the storage and release of gonadotropins by the pituitary is normal. If there was a successful stimulation of testicular T levels by hCG, but an inadequate or no response in LH production, then the patient has hypogonadotropic, secondary, hypogonadism.
In the simplest terms, the second half of the protocol is to determine hypothalamo-pituitary production and reserve with clomiphene and tamoxifen. The physiological type of hypogonadism—hypogonadotropic or secondary—is characterized by abnormal low or low normal gonadotropin (LH) production in response to clomiphene citrate and tamoxifen. In the functional type of hypogonadism, the ability to stimulate the HPTA to produce LH and T levels within the normal reference range occurs.
There is a dearth of good studies in anabolic steroids, both while you're taking them and after you stop them, I think this is going to be something that we're going to need to look at in the future. In fact, we are going to plan on looking at it in our proposed clinical studies that we have with our company for the prevention of anabolic steroid-induced hypogonadism.
Subscribe to Dr Scally's blog: http://michaelscally.blogspot.com/
More on HPTA reset
Attachments
Last edited: